Step 4: Where needed, place remaining electrons on the central atom: 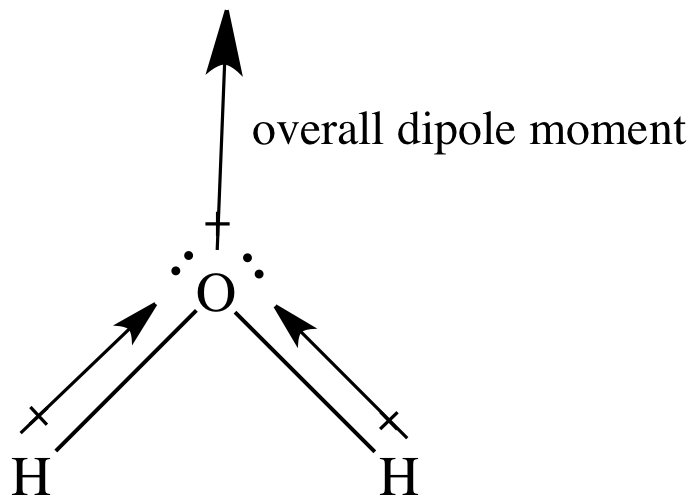
NH 3: no terminal atoms capable of accepting electrons HCCH: no terminal atoms capable of accepting electrons Step 3: Where needed, distribute electrons to the terminal atoms: 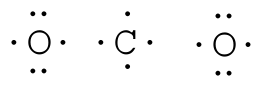
Draw a skeleton and connect the atoms with single bonds. Step 1: Calculate the number of valence electrons. Let us determine the Lewis structures of \text_(2), each atom has an octet as drawn, so nothing changes. Rearrange the electrons of the outer atoms to make multiple bonds with the central atom in order to obtain octets wherever possible.Place all remaining electrons on the central atom.Distribute the remaining electrons as lone pairs on the terminal atoms (except hydrogen), completing an octet around each atom.(Generally, the least electronegative element should be placed in the center.) Connect each atom to the central atom with a single bond (one electron pair). Draw a skeleton structure of the molecule or ion, arranging the atoms around a central atom.For anions, add one electron for each negative charge. For cations, subtract one electron for each positive charge. Determine the total number of valence (outer shell) electrons.The transition elements and inner transition elements also do not follow the octet rule:įor more complicated molecules and molecular ions, it is helpful to follow the step-by-step procedure outlined here: Because hydrogen only needs two electrons to fill its valence shell, it is an exception to the octet rule. These four electrons can be gained by forming four covalent bonds, as illustrated here for carbon in CCl 4 (carbon tetrachloride) and silicon in SiH 4 (silane). For example, each atom of a group 14 element has four electrons in its outermost shell and therefore requires four more electrons to reach an octet. The number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons) this is especially true of the nonmetals of the second period of the periodic table (C, N, O, and F). The tendency of main group atoms to form enough bonds to obtain eight valence electrons is known as the octet rule. This allows each halogen atom to have a noble gas electron configuration. The other halogen molecules (F 2, Br 2, I 2, and At 2) form bonds like those in the chlorine molecule: one single bond between atoms and three lone pairs of electrons per atom. Each Cl atom interacts with eight valence electrons: the six in the lone pairs and the two in the single bond. Inside the erythrocytes, the molecule involved in this crucial process, is 'hemoglobin', formed by four protein subunits and a heme group'.A single shared pair of electrons is called a single bond. Erythrocytes, commonly known as red blood cells are the cell type responsible for the gas exchange (i.e. Every process of protein formation, from the binding of individual amino acids to secondary structures to tertiary structures and even the formation of quaternary structures is dependent on dipole-dipole interactions.Ī prime example of quaternary dipole interaction that is vital to human health is the formation of erythrocytes. The biggest impact dipole interactions have on living organisms is seen with protein folding. The potential energy from dipole interactions is important for living organisms. \]īiological Importance of Dipole Interactions 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
